Automated SARS-CoV-2 Antibody Testing by ECLIA: What it is and what it is not
At the onset, it is important to know that antibody testing is not a test for the virus, it is not a molecular test like RT-PCR, and it is not the gold standard for diagnosis. Just like HIV testing for the AIDS pandemic, the development of antibody testing will have to undergo numerous improvements over the next several years.
Antibody testing

The Medical City offers ECLIA antibody test at its Drive-Thru COVID-19 testing site located at the parking area of the Ateneo School of Medicine and Public Health (ASMPH). The drive-Thru testing site is open Mondays to Sundays from 7AM to 6PM.For online booking, visit www.tmccovidtest.com.
Antibody testing is not a test for the virus. Instead, it is used to detect immunoglobulins (Ig) produced by a person in response to infections or other inciting agents. It is also called serologic testing because serum (liquid part of blood after the clot is separated) is historically the most frequent specimen used. Nowadays, testing may use plasma, whole blood, or even other body fluids.
There are many types and platforms of antibody testing. For purposes of the COVID-19 pandemic, let us simplify these into: manual and automated methods.
Manual testing commonly utilizes the lateral flow assay (LFA) or the so-called rapid antibody test kits (RATK). In ordinary times, stringent test validations are required before these kits are commercialized. Not surprisingly, many of these kits have been recalled by the US FDA starting last May 21.
Automated testing includes methods like enzyme-linked immunosorbent assay (ELISA) and chemiluminescence immunoassay (CLIA). These are well established methods and many tertiary clinical laboratories in the Philippines use this platform for many types of tests. It has a fast turn-around time in a machine that includes a tighter quality control compared to manual tests. Understandably, it eliminates the uncertainties involved in human visual evaluation (i.e. Is the faint line in the rapid test kit a negative or positive reading?).
Enhanced chemiluminescence immunoassay (ECLIA)
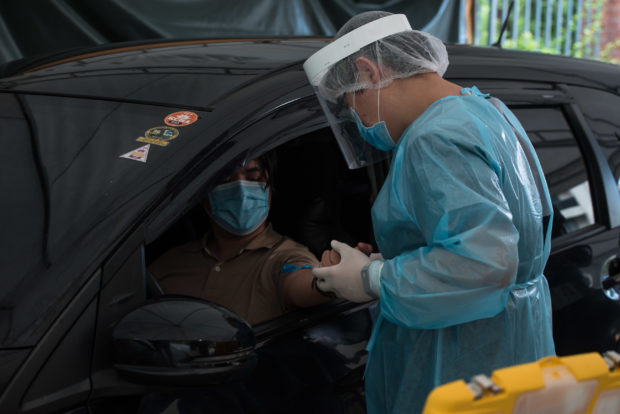
The Medical City offers ECLIA antibody test at its Drive-Thru COVID-19 testing site located at the parking area of the Ateneo School of Medicine and Public Health (ASMPH). The drive-Thru testing site is open Mondays to Sundays from 7AM to 6PM.For online booking, visit www.tmccovidtest.com.
ECLIA is not a molecular test like RT-PCR. It is a proprietary technology based on the principles of CLIA. The manufacturer is mandated to validate the test performance characteristics. Sensitivity refers to the percentage of patients who test positive out of all patients who have the disease (true positive rate). On the other hand, specificity refers to the percentage of patients who test negative out of all patients who do not have the disease (true negative rate). These test characteristics are estimates and should be used in the proper setting.
For example, the US FDA website provides this information on the Anti-SARS-CoV-2 CLIA Total Ig against the spike antigen: sensitivity 100% (CI: 92.7-100%) and specificity 100% (CI: 99-100%). CI stands for confidence interval which is the range that one is sure (usually at 95% confidence) that the test characteristics will fall within it. An extensive review on serodiagnostics for SARS-CoV-2 was recently published.
Aside from the validation performed by the manufacturer, verification must still be done by a clinical laboratory. This is intended to check if the test performs as well as, or better, than the claimed characteristics based on a set of criteria. Think of this as a test drive before a long distance road trip. This in-house verification data is kept as internal documents that may be reviewed by regulatory and accreditation bodies.
Timing of test
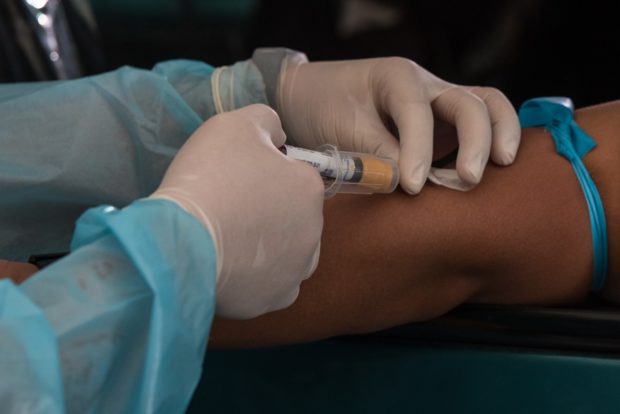
The Medical City offers ECLIA antibody test at its Drive-Thru COVID-19 testing site located at the parking area of the Ateneo School of Medicine and Public Health (ASMPH). The drive-thru testing site is open Mondays to Sundays from 7AM to 6PM.For online booking, visit www.tmccovidtest.com.
ECLIA is not the gold standard for diagnosis. In symptomatic patients, molecular testing remains as the mainstay for confirmation. However, it must be done at the right time, typically a week after exposure or a few days after onset of symptoms. Otherwise, a false negative result will be obtained. Think of this as taking a HIV test the next morning after an exposure.
Antibody testing must be done several days after viral exposure when the body has produced enough immunoglobulins (Ig) to be detectable by a test. For the total Ig test using ECLIA, the test performs best when done eight days after symptom onset. The combination of total Ig and IgG gives a better clinical assessment compared to serologic testing of total Ig alone or IgG alone.
The total Ig test detects all types of antibodies (IgM, IgG, IgA). Conventional knowledge has taught us that IgM is a marker of an early antibody response while IgG denotes a late response. However in COVID-19 infections, it has been shown that IgM appears at about the same time as IgG, thus, the rationale for total Ig testing.
Similar to other viral infections, IgG is present for several weeks during the convalescent phase, even in asymptomatic patients[4] . It is still not clear how long these IgG antibodies persist, or what kind of protection it provides. For now, think of this like HIV antibodies which are just markers of infection, until we have proof to the contrary.
As an added tool to assess patients, antibody testing may be considered in combination with RT-PCR since onset of community exposure may be difficult to ascertain. Those who wish to donate convalescent plasma would necessarily need to be tested. As with all diagnostic tests, it is best that a physician determine if and when a test is needed, while knowing the explanation for the different result combinations in the context of the clinical history and other findings.
Antibody testing for HIV has gone a long way, yet it remains imperfect in spite of the knowledge after four decades. We still need to learn a lot about this novel virus four months into the pandemic.
Right now, we can be guided by this dictum: RIGHT TEST using the RIGHT METHOD at the RIGHT TIME.
This article was written by Arvin C. Faundo, M.D., consultant director for Immunology, Department of Laboratory Medicine and Pathology, The Medical City.
For inquiries about ECLIA antibody testing at The Medical City, you may call 89881000 or 89887000.